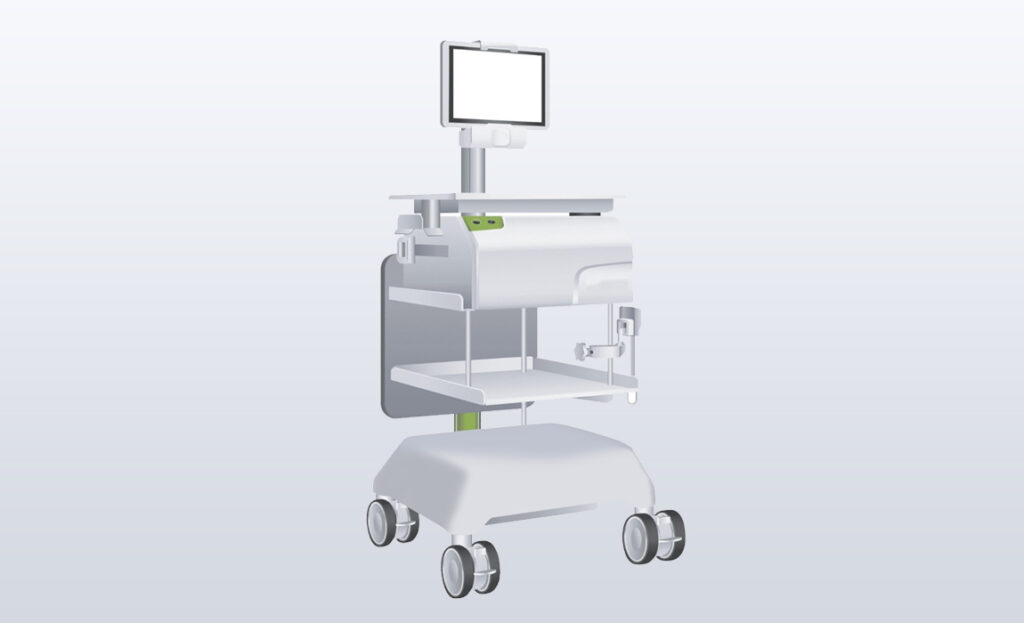
Overview
The customer was preparing for market launch of a disruptive medical device, but realized they had a workflow problem in the OR that could negatively impact commercialization. They had committed to investors that their FDA 510(k) application would be complete within 4 months. The oversight in workflow jeopardized their regulatory milestones, market launch, and investor covenant.
Process
MPE integrated a collection of technology into an efficient, mobile platform. Produced design, technical drawings, and physical product for FDA filing in 4 months. Product passed all regulatory and safety testing requirements.
Solution
Our customer made good on commitments to investors and subsequently exceeded their venture capital funding targets, ensuring an accelerated commercial launch. FDA clearance achieve on first pass. Product is in the market and is streamlining treatment pathways and improving outcomes for women diagnosed with breast cancer.
Increase Revenue
Increase revenue by exactly matching market needs without compromise in a timely manner. Increase brand equity by consistency and coherence across the offering.
Lower Cost
Quickly and cost-efficiently bring custom products to market with significant experience in the MedTech industry. Lower costs realized by efficiencies in the product development process (i.e., ID to DE to PE to Sustaining).
Reduce operational TCO via supplier consolidation & scale, reduced lead times, warehousing/inventory management, and 3PL services.
Lower Risk
Years of experience help avoid inadvertent regulatory hurdles typically associated with custom solutions.